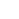Article Source : CISION PR Newswire
SEOUL, South Korea, March 28, 2022 /PRNewswire/ — AI-based digital mental healthcare platform company iMediSync has announced that their product “iSyncWave” has won the 2022 Red Dot Design Award for its innovative and creative design as a dry EEG headset.
Hosted by Germany’s Design Zentrum Nordheim Westfalen, the Red Dot is considered one of the three most prestigious design awards in the world, standing tall with iF, IDEA Awards.

The next generation dry EEG measurement device, iSyncWave
iSyncWave offers a high-quality EEG/HRV measurement and scanning technology that has been applied in multiple clinical scenes including hospitals, research facilities, and Alzheimer’s Treatment Center in South Korea, and continues close cooperation with an overseas brain development center, sports club, and rehabilitation center. The product displays its efficiency and utility in the various fields of neurology and psychology, allowing the iSyncWave to become the first QEEG headset product to win the Red Dot Design Award in South Korea.
The CEO of iMediSync, Seungwan Kang, says “iSyncWave provides a QEEG and an HRV measurement technology and an analysis report based on the over 1,300 healthy brainwave databases covering all ages. Also, an AI automatic analysis cloud technology, a biomarker that pre-detects any signs of aMCI in the early stages.The Red Dot Design Award has proved the product’s innovation and creativity, efficiency to show its effect not only in the clinical field but also user-friendly services and a platform that the clients themselves had experienced, highlighting the company’s effort and the vision to provide the best service to the clients as the leading Tele-Medicare platform which we are proud of”.
In addition, iMediSync is continuing their joint research with the MOU partners for developing their pipeline for a biomarker, including the detection of coma, post-aMCI symptoms for stroke to set themselves as a global standard for a digital mental healthcare platform service.
Recently, iMediSyncd has acquired a Korean FDA and a U.S. FCC approval for their product iSyncWave and is looking to clear the U.S. FDA approval this May, which the company expects will boost their global market expansion and lead a trend as digital mental health care in the post-covid era that opens a whole new possibility for an untact Tele-Medicare platform.